Magnetic resonance imaging in patients with spinal neurostimulation systems. After the exam, MRI-mode should be turned off and settings returned to pre-MRI values. ĭe Andres J, Valía JC, Cerda‐Olmedo G et al. Visual and auditory monitoring should be continuously performed during the scan. The patient must be in a cognitive state able to provide immediate feedback concerning any problems during the examination. For several models there are restrictions on the use of transmit body coils and power deposition limits dependent on the location of magnet isocenter. 
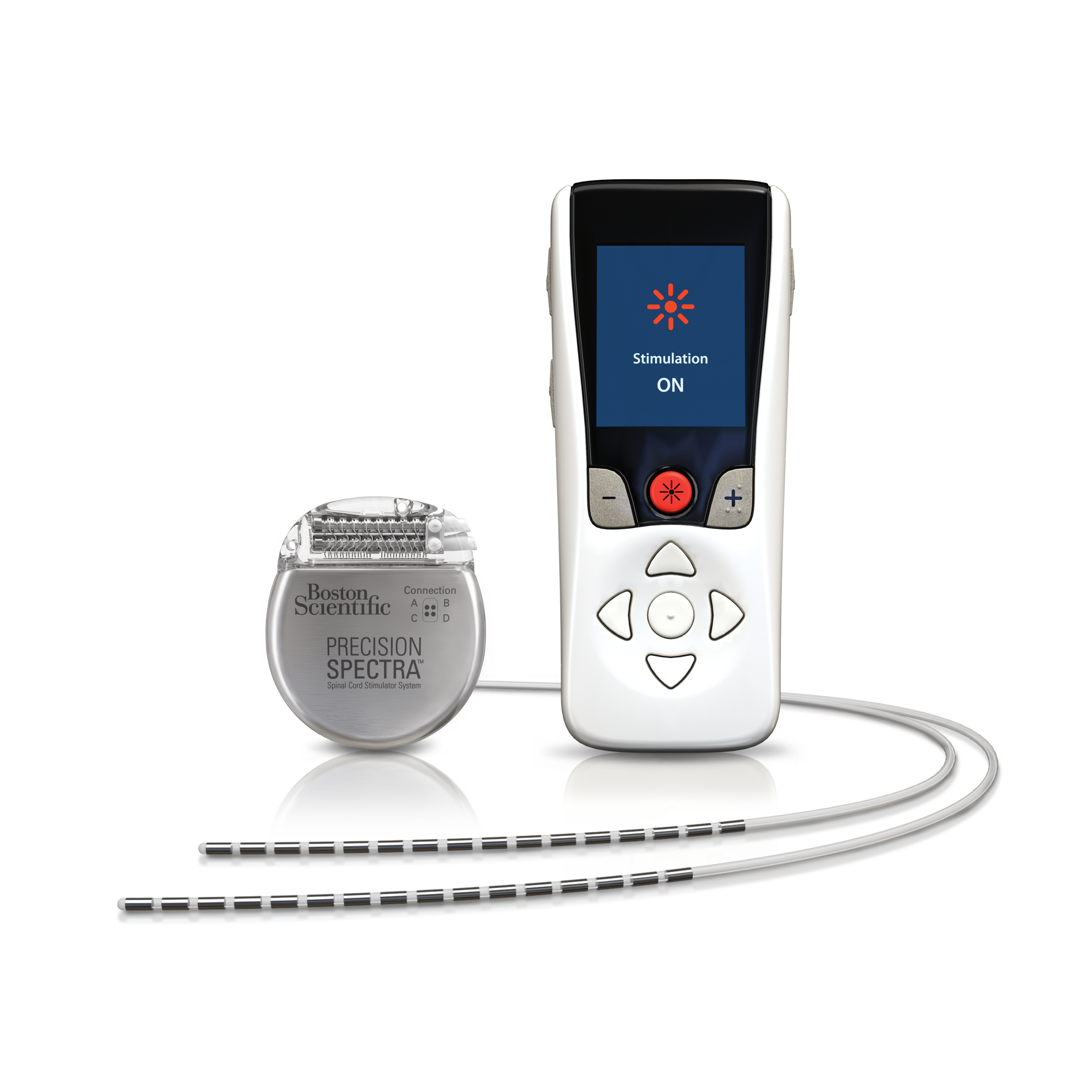
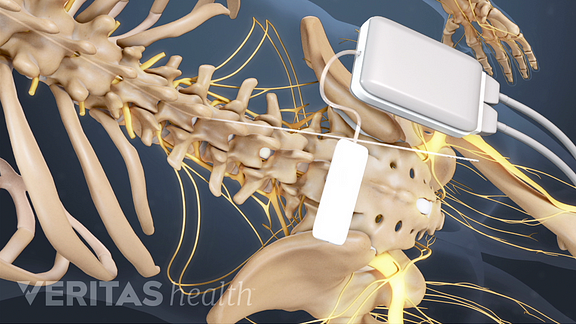
Limits for maximum spatial gradient, gradient slew rate, total imaging time, and RF power dissipation measured by average SAR or maximum B 1+RMS must be followed explicitly. These conditions will vary significantly between manufacturers and SCS models. All current MR-conditional SCS devices are restricted to ¹H imaging in horizontal bore cylindrical magnets operating at 1.5 or 3.0T. Only the supine or prone patient positions are allowed. The IPG should be turned off or placed in "MRI Mode" if available. The IPG battery should be fully charged and lead impedances should measure within expected values. Pre-MRI evaluation/interrogation of the DBS system.Insure that the wires are connected to the IPG without breaks and there are no abandoned leads or electrodes. Verify (by x-ray if necessary) that the IPG has been implanted subcutaneously in the pelvis/flank. This can be done by reviewing operative notes from the medical record or the patient's information card. The exact model/serial numbers from the various components of the SCS system, including the IPG, leads, and extensions (if present) must be established with certainty. Presented at: 37th Annual Meeting of the North American Spine Society, 2022. Surgical treatment of refractory low back pain using implanted BurstDR spinal cord stimulation (SCS) in a cohort of patients without options for corrective surgery: findings and results from the DISTINCT study, a prospective randomized multi-center controlled trial. Abbott recieves FDA approval for its spinal cord stimulation systems to treat chronic back pain in people who have limited surgical options. "This FDA expanded indication approval for our SCS devices is a significant step forward in Abbott's goal to provide treatment access to those who suffer daily with chronic back pain but are not eligible for corrective surgery,” Pedro Malha, vice president, Neuromodulation, Abbott, said in a statement. Pain-related emotional distress was also improved in the SCS treatment arm in relation to CMM. Those on SCS continued to report superior meaningful reductions in pain intensity and low back pain-related disability compared with CMM. Following the trial, 125 percutaneous and 50 paddle leads were implanted, most (96%) of which were between T7 and T9. After the 6-month trial period, 87% of patients reported at least 50% pain relief and NRS scores for low back pain decreased from 7.7 to 2.5. 1 "This new indication for Abbott's SCS devices, together with BurstDR stimulation, allows physicians the ability to identify and treat a new group of people, providing them with relief from chronic back pain."Īdditional findings from DISTINCT were presented at the 37th Annual Meeting of the North American Spine Society in 2022, further supporting the efficacy of the SCS systems. "To date, we have struggled with how to treat people who weren't considered a good surgical candidate because we didn't have clear, data-driven treatment options for non-surgical back pain,” Timothy Deer, MD, FIPP, DABPM, president and chief executive officer, Spine and Nerve Centers of the Virginias, Charleston, West Virginia, said in a statement. ON average, people who received SCS therapy experienced a 69.7% reduction in pain. In the study, 91.4% of people who received SCS therapy obtained significant pain relief or significantly improved function. Comprised of 270 individuals who suffered an average of 12.8 years with pain, 72.6% of those in the SCS study arm and 85.2% of those implanted achieved significant back pain reduction compared with only 7.1% of individuals in the conventional medical management (CMM) arm.Īll of Abbott’s SCS therapies in the US feature the company’s proprietary, low-energy BurstDR waveform, which had efficacy further supported by DISTINCT. The approval was based on the DISTINCT study (NCT0447987), the largest randomized controlled trial for SCS in people with chronic back pain for which surgery is not an option. The new indication spans across the entire SCS program, which includes the recharge-free Proclaim SCS family and the rechargeable Eterna SCS platform. According to an announcement, the FDA has approved Abbott’s spinal cord stimulation (SCS) devices for the treatment of chronic back pain in individuals who have not or are not eligible to receive back surgery. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
